Read and download the CBSE Class 10 Chemistry Periodic Classification Of Elements Worksheet Set 03 in PDF format. We have provided exhaustive and printable Class 10 Science worksheets for Chapter 5 Periodic Classification of Elements, designed by expert teachers. These resources align with the 2026-27 syllabus and examination patterns issued by NCERT, CBSE, and KVS, helping students master all important chapter topics.
Chapter-wise Worksheet for Class 10 Science Chapter 5 Periodic Classification of Elements
Students of Class 10 should use this Science practice paper to check their understanding of Chapter 5 Periodic Classification of Elements as it includes essential problems and detailed solutions. Regular self-testing with these will help you achieve higher marks in your school tests and final examinations.
Class 10 Science Chapter 5 Periodic Classification of Elements Worksheet with Answers
Multiple choice questions
Question. According to Mendeleev’s Periodic Law, the elements were arranged in the periodic table in the order of
(a) increasing atomic number
(b) decreasing atomic number
(c) increasing atomic masses
(d) decreasing atomic masses
Answer. C
Question. Which of the following statement(s) about the Modern Periodic Table are incorrect
(i) The elements in the Modern Periodic Table are arranged on the basis of their decreasing atomic number
(ii) The elements in the Modern Periodic Table are arranged on the basis of their increasing atomic masses
(iii) Isotopes are placed in adjoining group (s) in the Periodic Table
(iv) The elements in the Modern Periodic Table are arranged on the basis of their increasing atomic number
(a) (i) only
(b) (i), (ii) and (iii)
(c) (i), (ii) and (iv)
(d) (iv) only
Answer. B
Question. On the basis of electronic configuration of 5X, the group number and period of the element ‘X’ is:
(a) Group 15 period 2
(b) Group 13 period 2
(c) Group 19 period 5
(d) Group 13 period 5
Answer. B
Question. An element ‘X’ is forming an acidic oxide. Its position in modern periodic table will be
(a) Group 1 and Period 3
(b) Group 2 and Period 3
(c) Group 13 and Period 3
(d) Group 16 and Period 3
Answer. D
Question. Elements P, Q, R and S have atomic numbers 11, 15, 17 and 18 respectively. Which of them are reactive non-metals?
(a) P and Q
(b) P and R
(c) Q and R
(d) R and S
Answer. B
Question. Which of the following elements has 2 shells and both are completely filled?
(a) Helium
(b) Neon
(c) Calcium
(d) Boron
Answer. D
Question. Which of the following are the characteristics of isotopes of an element?
(i) Isotopes of an element have same atomic masses
(ii) Isotopes of an element have same atomic number
(iii) Isotopes of an element show same physical properties
(iv) Isotopes of an element show same chemical properties
(a) (i), (iii) and (iv)
(b) (ii), (iii) and (iv)
(c) (ii) and (iii)
(d) (ii) and (iv)
Answer. C
Question. Three elements B, Si and Ge are
(a) metals
(b) non-metals
(c) metalloids
(d) metal, non-metal and metalloid respectively
Answer. C
Question. Which of the given elements A, B, C, D and E with atomic number 2, 3, 7, 10 and 30 respectively belong to the same period?
(a) A, B, C
(b) B, C, D
(c) A, D, E
(d) B, D, E
Answer. B
Question. Where would you locate the element with electronic configuration 2, 8 in the Modern Periodic Table?
(a) Group 8
(b) Group 2
(c) Group 18
(d) Group 10
Answer. C
Question. Newlands relation is called
(a) Musical Law
(b) Law of Octaves
(c) Periodic Law
(d) Atomic Mass Law
Answer. B
Today, 118 elements are known, the first 94 of which occur in nature. Of the 94 natural elements, eighty are stable.The periodic table is a graphic description of the periodic law, which states that the properties and atomic structures of the chemical elements are a periodic function of their atomic number.
Elements are placed in the periodic table by their electron configurations, which exhibit periodic recurrences that explain the trends of properties across the periodic table. As we go across a period from left to right, we add a proton to the nucleus and an electron to the valence shell with each successive element. As we go down the elements in a group, the number of electrons in the valence shell remains constant, but the principal quantum number increases by one each time.
An understanding of the electronic structure of the elements allows us to examine some of the properties that govern their chemical behavior. These properties vary periodically as the electronic structure of the elements changes.
They are
(1) size (radius) of atoms and ions,
(2) ionization energies, and
(3) electron affinities.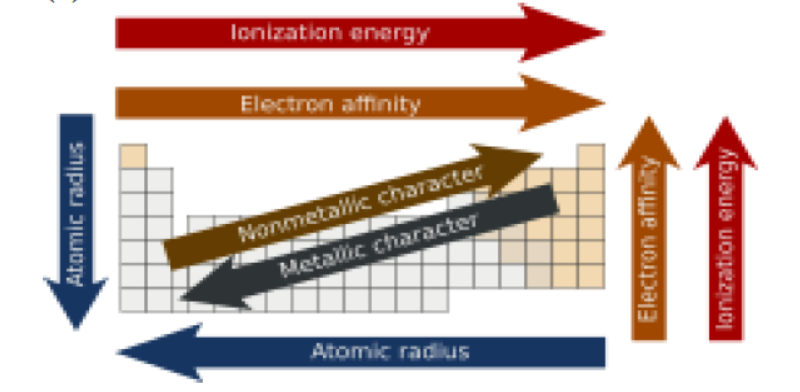
Question. Which of the following set of elements is written in order of their increasing metallic character?
(a) Na, Li, K
(b) C, O, N
(c) Mg, Al, Si
(d) Be, Mg, Ca
Answer. D
Question. Which of the following is the correct order of the atomic radii of the elements oxygen, fluorine and nitrogen?
(a) O < F < N
(b) N < F < O
(c) O < N < F
(d) F < O < N
Answer. D
Question. The positions of four elements A, B, C and D in the modern periodic table are shown below. Which element is most likely to form an acidic oxide?
(a) A
(b) B
(c) C
(d) D
Answer : C
Question. Which of the following elements would lose an electron easily?
(a) Mg
(b) Na
(c) K
(d) Ca
Answer : C
Question. Which of the following elements does not lose an electron easily?
(a) Na
(b) F
(c) Mg
(d) Al
Answer : B
Question. Which among the following elements has the largest atomic radii?
(a) Na
(b) Mg
(c) K
(d) Ca
Answer : C
Question. Elements P, Q, R and S have atomic numbers 11, 15, 17 and 18 respectively. Which of them are reactive non-metals?
(a) P and Q
(b) P and R
(c) Q and R
(d) R and S
Answer : C
Question. Which of the following gives the correct increasing order of the atomic radii of O, F and N?
(a) O < F < N
(b) N < F < O
(c) O < N < F
(d) F < O < N
Answer : D
Question. What happens to tendency to gain electron in a period?
(a) Increases,
(b) Decreases,
(c) Remaining same,
(d) First increases then decreases.
Answer. A
Question. Which of the following elements would lose an electron easily?
(a) Mg
(b) Na
(c) K
(d) Ca
Answer. C
Question. Atomic size decreases from left to right in a period because
(a) Effective nuclear charge increases
(b) Number of shells remains the same
(c) Force of attraction between the nucleus and valence electrons increases
(d) All of these
Answer. D
Assertion-Reason Type Questions
For question numbers 1 to 2 two statements are given-one labeled as Assertion (A) and the other labeled
Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below:
(a) Both ‘A’ and ‘R’ are true and ‘R’ is correct explanation of the assertion.
(b) Both ‘A’ and ‘R’ are true but ‘R’ is not correct explanation of the assertion.
(c) ‘A’ is true but ‘R’ is false.
(d) ‘A’ is false but ‘R’ is true.
Question. Assertion: X with atomic number 13 is a metal.
Reason: It belongs to group 13 and 3rd period.
Answer : B
Question. Assertion: Carbon is a metalloid.
Reason: Carbon forms CO2 which is acidic oxide whereas CO is neutral oxide.
Answer : D
Very Short Answer Type Questions
Question. If Lithium, sodium and potassium form a Dobereiner’s triad, and if the atomic masses of Li and K are 7 and 39, respectively, Predict the atomic mass of sodium?
Answer : Sum of atomic masses of extreme elements=7+39=46. Average at mass =46/2 =23.So sodium will have atomic mass =23
Question. Name the two elements for which temporary names were given as Eka-aluminium and Eka-silicon and spaces were left by Mendeleev in his table even before their discovery.
Answer : Gallium and Germanium
Question. Write the formulae of chlorides of Eka-silicon and Eka-aluminium, the elements predicted by Mendeleev.
Answer : Formula of Chloride of Eka Silicon is GeCl4 and that of Eka Aluminium is GaCl3
Question. State Mendeleev periodic law
Answer : Properties of elements are a periodic function of their atomic masses.
Question. How does atomic size vary from left to right in a period?
Answer : Atomic size decreases from left to right in a period.
Question. Which has smaller size: K(19) or Na(11); B(5) or C(6)?
Answer : Na(11) is smaller in size than K(19), C(6) is smaller in size than B(5).
Question. On moving from left to right in the second period, what happens to the number of valence electrons?
Answer : Valence electrons keeps on increasing from left to right in the second period.
Question. Write the number of valence electrons present in a nitrogen atom (714N)
Answer : It has 5 valence electrons.
Question. How does atomic size vary from left to right in a periodic table?
Answer : Atomic size decreases along a period from left to right in the periodic table.
Question. How does metallic character (electropositive character) varies down the group?
Answer : It increases down the group.
Question. Give any one difference in the electronic configuration of Group 1 and Group 2 elements.
Answer : Group 1 elements have 1 valence electron and are more reactive than Group 2 elements which have two valence electrons.
Question. The formula of magnesium oxide is MgO. State the formula of barium nitrate and barium sulphate, if barium belongs to the same group.
Answer : Ba(NO3)2, BaSO4
Question. “Fluorine is more electronegative than iodine”. Give reason in support of this.
Answer : ‘F’ is smaller in size than I, therefore the tendency to gain electrons is more due to more effective nuclear charge.
Question. What would be nature of oxides formed by the elements on the right hand side of periodic table?
Answer : Acidic
Question. The electronic configuration of two elements ‘A’ and ‘B’ are 2, 8, 7 and 2, 8, 8, 2, respectively. Write the atomic number of these elements. What will be the formula of the compound formed and the nature of bond between them, when these elements chemically combine together?
Answer : A has atomic number ‘17’, ‘B’ has atomic number ‘20’.
BA2 is the formula of the compound. The bond formed between A and B will be ionic bond
Question. Which has larger atomic radius, K(19) or Ca(20)?
Answer : K(19) is larger than Ca(20).
Question. How does reactivity of metals vary down the group?
Answer : It increases down the group.
Question. Define electropositivity.
Answer : It is defined as measure of tendency to lose electrons. The greater the tendency to lose electrons, more will be electropositivity.
Question. Which is smaller: (i) Na+ or Na, (ii) Cl or Cl– ?
Answer : (i) Na+, (ii) Cl
Question. Arrange the following metals in decreasing order of atomic size: Ca, Mg, Ba, Be
Answer : Ba > Ca > Mg > Be
Question. List any two properties of the elements belonging to the first group of modern periodic table.
Answer : (i) They should have valency equal to 1 and form monovalent positive ions.
(ii) They are highly reactive soft metals
Question. How does valency of an element vary across a period?
Answer :The valency of an element first increases and then decreases across a period.
Question. Out of Li and K, which one have stronger metallic character and why?
Answer :K’, because it can lose electrons easily due to larger size and less effective nuclear charge.
Question. The atomic radii of first group elements are given below:
Group-1 element Atomic Radii (pm)
Na 86
K 231
Rb 244
Cs 282
State the reason behind the observed trend in the above elements.
Answer : Atomic radii increases down the group because number of shells go on increasing, effective nuclear charge decreases, distance between nucleus and valence shell increases
| Chapter 16 Digestion and Absorption Printable Worksheet |
CBSE Science Class 10 Chapter 5 Periodic Classification of Elements Worksheet
Students can use the practice questions and answers provided above for Chapter 5 Periodic Classification of Elements to prepare for their upcoming school tests. This resource is designed by expert teachers as per the latest 2026 syllabus released by CBSE for Class 10. We suggest that Class 10 students solve these questions daily for a strong foundation in Science.
Chapter 5 Periodic Classification of Elements Solutions & NCERT Alignment
Our expert teachers have referred to the latest NCERT book for Class 10 Science to create these exercises. After solving the questions you should compare your answers with our detailed solutions as they have been designed by expert teachers. You will understand the correct way to write answers for the CBSE exams. You can also see above MCQ questions for Science to cover every important topic in the chapter.
Class 10 Exam Preparation Strategy
Regular practice of this Class 10 Science study material helps you to be familiar with the most regularly asked exam topics. If you find any topic in Chapter 5 Periodic Classification of Elements difficult then you can refer to our NCERT solutions for Class 10 Science. All revision sheets and printable assignments on studiestoday.com are free and updated to help students get better scores in their school examinations.
FAQs
You can download the latest chapter-wise printable worksheets for Class 10 Science Chapter Chapter 5 Periodic Classification of Elements for free from StudiesToday.com. These have been made as per the latest CBSE curriculum for this academic year.
Yes, Class 10 Science worksheets for Chapter Chapter 5 Periodic Classification of Elements focus on activity-based learning and also competency-style questions. This helps students to apply theoretical knowledge to practical scenarios.
Yes, we have provided solved worksheets for Class 10 Science Chapter Chapter 5 Periodic Classification of Elements to help students verify their answers instantly.
Yes, our Class 10 Science test sheets are mobile-friendly PDFs and can be printed by teachers for classroom.
For Chapter Chapter 5 Periodic Classification of Elements, regular practice with our worksheets will improve question-handling speed and help students understand all technical terms and diagrams.

