Important Points
A Organic compounds containing oxygen - I
1. Preparation of alcohols:
→ Monohydric alcohols are prepared by the hydrolysis of alkyl halides with aqueous alkali, hydration of alkenes, hydrolysis of ester, reduction of (alde hydes, ketones, acids and acid derivatives). Grignard reagents is also used to prepare monohydric alcohols.
2. Physical properties of alcohols:
→ The boiling points of alcohols are much higher than comperatively same molecular masses of alkanes, ethers and alkyl halides. This is due to intermo lecular Hbond. For isomeric alcohols, the boiling points are in the order 10>20>30.
→ Due to the formation of H-bond between alcohol and H2O modecules, alcohol with lower number of carbons are soluble in water.
3. Chemical properties of alcohols:
→ Alcohols exhibit three types of reactions,
(i) Reaction in which O-H bond cleaves
(ii) Reaction in which C-O bond cleaves
(iii) Reaction in which whole molecule of alcohol participate.
→ Victor-Meyer’s test and Lucas reagent are used to distinduish 1°, 2° and 3° alcohols.Oxidation reactions are also used to distiguished between 1°, 2° and 3° alcohols.
4. Preparation of phenol:
→ Phenol is prepare from cumene, diazonium salt, benzene and coal tar.
5. Physical properties of phonol:
→ Phenols have higher boiling point than the corresponding hydrocarbon and aryl halides. This is due to the presence of intermolecular hydrogen bonding.
→ Phenols are more acidic than alcohols because phenoxide ion is stabilised by resonance. The presence of electon withdrawing group like NO2, increases the acidic strength of phenol and electron donating group like R, decreases the acidic strength of phenol.
6. Chemical properties of phenol:
→ Reaction of phenols are mainly of two types,
(i) Reaction involving OH group
(ii) Reaction involving phenyl group
7. Preparation of ethers:
→ Ethers are mainly prepared by Williamson’s synthesis which involces the heating of alkyl halides with sodium or potassium alkoxides or phenoxides. Dehydration of alcohol at 1400c also gives ether.
8. Chemical properties of ethers:
→ (i) Reaction involving cleavage of C-O bond with dilute acid or with HX
(ii) Electrophillic substitution reactions occuring in aromatic ring
B: Organic compounds containing oxygen - II
1. Carbonyl compounds:
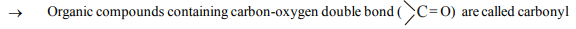
group or carboxy group compounds. In aldehydes, the carbonyl group is attached to one hydrogen atom and one alkyl (or aryl or hydrogen atom) group, while in ketones it is attached to one alkyl and one arly group or to two alkyl (or aryl) groups, which may be same or diffrent. If carbonyl group is attached to one hydroxyl group, the compounds are know as carboxylic acids. In carboxylic acid compounds, if the hydrogen of hydroxyl group is substituted by alkyl or aryl group the compounds are known as esters, but if it is substitued by acyl group, the compounds are known as acid anhydrides. If the carbonyl group is attached to chlorine and to amino group the compounds are known as acid chlorides and amides respectively. The general formula of these compounds are expressed as Amide
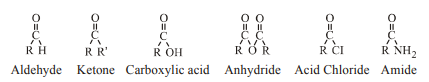
2. Structure and nature of carbonyl group:
→ Carbonyl carbon atom is sp2 hybridised and form three σ-bonds and one ∏ bond. All the three s-bond lie in same plane having angle 1200. The ∏- bond lies both above and below the C-O s bond. Thus the carbonyl carbon, oxygen atom and two atoms which are directly bonded to the carbonyl carbon lie in one plane, and is confirmed by electron diffraction and spectroscopic studies.
→ Due to higher electronegativity of oxygen atom relative to carbon atom the carbonyl group is polarized and carbonyl carbon becomes electrophile (Lewis acid) and oxygen becomes nucleophile (Lewis base). Carbonyl group is polar in nature and has dipole moments.Aldehydes and ketones have dipole moments 2.3-2.8 D. The resonance structures are as shown below :
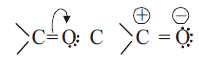
3. Physical properties of aldehydes and ketones:
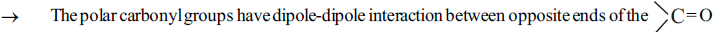
group dipoles and hence due to weak intermolecular attraction the melting points and boiling points of aldehydes and ketones are higher than corresponding non-polar compounds.
The order of boiling points is carboxylic acid > alcohol > isomeric ketone > isomeric aldehyde > ether > hydrocarbon.
→ Due to hydrogen bonding with water molecules the aldehydes and ketones upto three carbon are soluble in water.
The aromatic aldehydes and ketones due to presence of larger hydrocarbon parts (like benzenering etc.), are insoluble in water.
All adehydes and ketones are fairly soluble in organic solvents like benzene, ether, alcohols,chloroform etc.
4. Chemical properties of aldehydes and ketones:
→ Due to presence of hydrogen atom, the carbonyl group of aldehyde is much more reactive than ketone.
Aldehydes and ketones undergo nucleophilic addition reaction because the carbonyl carbon atom is slightly positively charged.
In nucleophilic addition the first step is reversible and also slow, so it is a rate determining step.
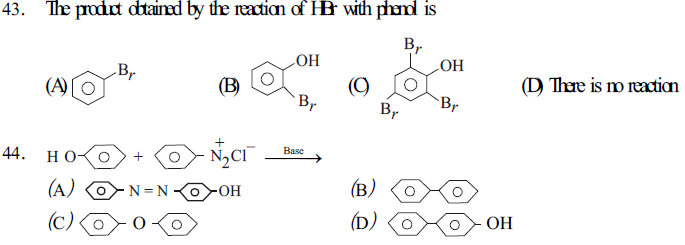
The second step is reversible.
Due to steric effect and inductive effect the aldehydes are more reactive than ketones.
Most of the aldehydes and aliphatic methyl ketones, due to less steric hindrance are more reactive.
→ Aldehydes and ketones react with NaHSO3 and give bisulphite addition product which are usually crystalline solids. On hydrolysis they give original aldehydes and ketones, so this reaction is useful for separation and purification of aldehydes and ketones.
Addition of HCN and Grignard reagent to the aldehyde and ketone which give α -hydroxy carboxylic acid and 1° , 2° ,3° alcohols respectively.
Addition of alcohol to aldehyde give hemiacetal and further acetal, while ketone give the same product.
Nucleophilic addition reaction of aldehydes and ketones with 3 NH and its derivatives (H2N - Z) are catalysed by acids.
→ Aldehydes and ketones on reduction give 1° and 2° alcohols respectively.
Aldehydes and ketones can be reduced to hydrocarbon by using different reagent like Wolff- Kishner reduction, Clemmenses reduction, red phosphorus with HI. Ketones on reduction with magnesium amalgam and water give the product pinacol. Oxidation of aldehydes : Tollens’ test, Fehling’s test and Benedict’s test give the product carboxylic acid. Fehling’s test and Benedict’s test are not given by aromatic aldehydes.
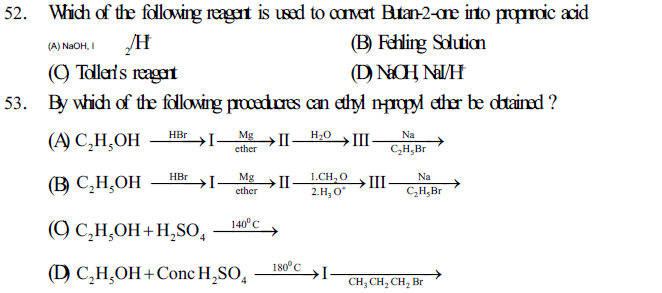
Oxidation of ketones by strong oxidizing agents like con. HNO3, KMnO4/ H2SO4, K2Cr2O7/H2SO4 give mixture of carboxylic acids.
Oxidation of aldehydes and ketones containing CH3CO -group give iodoform test.
→ Aldol condensation and cross aldol condensation are the reactions given by aldehydes and ketones having α -hydrogen atom or atoms using dilute alkali as catalyst.
Cannizzaro reaction is given by aldehydes and ketones which do not have an α -hydrogen atom by using con. NaOH or 50 % NaOH.
Electrophilic substitution reactions of aromatic aldehydes and ketones are nitration, sulphonation and halogenation.
5. Preparation of caroxylic acids :
Carboxylic acids are prepared from :
• Primary alcohol and aldehyde
• Alkyl benzene and alkenes
• Nitriles and amides.
• Grignard reagents
• Acid halide (chloride) and anhydrides
• Esters
6. Acidic nature of carboxylic acids :
→ Carboxylic acids are stronger acids than phenol and alcohols.
For convenience the strength of an acid is generally indicated by its a pKa value rather than its Ka value.
pKa = –log Ka
Factors affecting strength of acids are
• effect of electron-donating group
• effect of electron withdrawing group
• attachment of phenyl or vinyl group directly to carbonyl group.
7. Physical and chemical properties of carboxylic acids :
→ Carboxylic acid in aqueous solution form intermolecular hydrogen bonding with water molecules.
Carboxylic acids are cyclic dimer in vapour phase or in aprotic solvents.
→ The reactions of carboxylic acid are
• Reactions involving cleavage of O-H bond.
• Reactions involving cleavage of C-OH bond
• Reaction involving -COOH group
Substitution reaction in hydrocarbon part of carboxylic acid are halogenation and ring substitution
as bromination, nitration and sulphonation.
Carboxylic acids are used in different fields.
MCQ
1. Numbers of isomeric alcohols of molecular formula C5H12O are
(A) 5 (B) 8 (C) 6 (D) 9
2. Which of the following will produce only one product on reduction with LiAlH4 ?
(A) CH3COOCH2CH3 (B) CH3CH2OCOCH2CH3
(C) CH3COOCH3 (D) CH3CH2OCOCH2CH2CH3
6. Acid catalysed hydration of alkenes except ethene leads to the formation of
(A) primary alcohal
(B) mixture of primary and secondary alcohols
(C) secondary or tertiary alcohol
(D) mixture of secondary and tertiary alcohols
7. During dehydration of alcohol to alkenes by heating with Conc H2SO4, the initiation step is:
(A) elimination of water (B) formation of an ester
(C) formaton of carbocation (D) protonation of alcohol molecule
8. which of the following compounds will give positive iodoform test ?
(I) 3- methyl propan-2-ol (III) I - methyl cyclopentanal
(II) I - phenyl propan-1-ol (Iv) 3- phenyl propan-2-ol.
(A) I and III (B) I and IV (C) II and III (D) II and IV
9. Which of the following alcohal on heating with conc H2SO4 gives product, which show geometrical isomerism ?
(A) 2,4- dimethyl pentan-3-ol B) 2- methyl butan -2-ol
(C) butan-2-ol (D) all of the above
10. Propan -l-ol and propan-2-ol can be distingaished by
(A) oxidation with alkaline KMnO4 followed by reation with Fehing Solution
(B) oxidation with acidic dichromate followed by reation with Fehling Solution
(C) oxidation by heating with copper followed by reation with Fehling Solution
(D) oxidation with conc H2SO4 followed by reaction with Fehling Solution
11. Which one of the follwing will most readily be dehydrated in acidic condition ?
12. The best reagent to convert pent - 3 - en-2-ol into pent-3en-2-one is
(A) acidic permanganate (B) acidic dichromate
(C) chromic anhydride in glacial acetic acid (D) pyridinium chlorochromate
13. CH3 CH2 OH can not be prepare by which of the following reaction ?
(A) Hydrolysis of ethyl acetate
(B) Hydroboration of ethene followed by oxidation in basic medium
(C) Reaction of ethyl chloride with alcoholic potassium hydroxide
(D) Reaction of ethylacetate
14. The most suitable reagent for the conversion of primary alcohol into aldehyde with the same number of carban is
(A) acidified K2Cr2O7 (B) alkaline KMnO4
(C) acidified KMnO4 (D) pyridinium chlorochromate
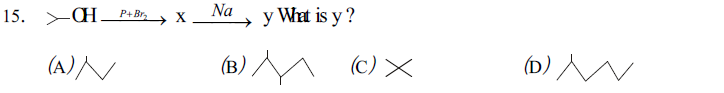
16. An organic compound "X" on treatment with PDC in CH2 Cl2 gives compound "Y". Compound "Y", reacts with I2 and alkali to form yellow precipitate. The compound "X" is
(A) acetone (B) ethanal (C) ethanol (D) acetic acid.
17. How many optically active stereoisomers are possidle for butan -2,3 -diol ?
(A) 1 (B) 2 (C) 3 (D) 4

19. The boiling point of glycerol is more than propanol because of
(A) hydrogen bonding (B) hybridization
(C) arrangment of molecules (D) size of molecule
20. An organic compound X is oxidised by using acidified 2 2 7 K Cr O . The product obtained reacts with phenyl hydrazine but does not give silver mirror test. The possible structure of X is
(A) CH3 CH2 OH (B) CH3 CO CH3
(C) ( CH3)2 CH OH (D) CH3 CHO
21. ( CH3)3 MgCl on reaction with D2O produces
(A) ( CD)3 CD CD (B) ( CD3)3 CH (C) ( CH3)3 COD (D) ( CH3)3 CD
22. Lucas test is associated with
(A) alcohols (B) phenols (C) aldehydes (D) carboxylic acids
23. ______________alcohol reacts immediately with anhydrous ZnCl2 + HCl and gives insoluble chloride
(A) Methanol (B) Ethanol
(C) Isopropyl alcohol (D) 2 - Methyl propan - 2-ol,
24. Glycerol is more viscous than ethanol due to
(A) many hydrogen bonds per molecule (B) high boiling point
(C) high molecular weight (D) Fajan's rule
25. 4.6 gram ethanol when reacts with sodium metal___________is formed.
(A) 11.2 litre H2 at STP (B) 1.12 litre H2 at STP
(C) 1.12 litre O2 at STP (D) 11.2 litre H2 at STP

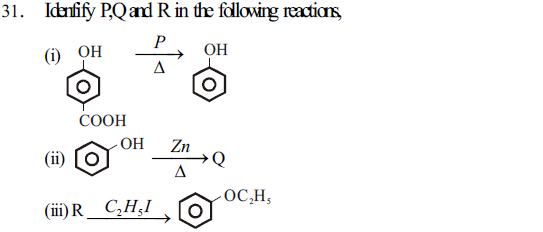
(A) Sodium oxide benzene, Sodium phenoxide
(B) Sodalime, benzene, potassium phenoxide
(C) Zn, cyclohexanone, Sodium ethoxide
(D) Sodium, cyclohexanone, potassium benzoate
32. Salicyladehyde and o-nitrophenol are less soluble in water because,
(A) - CHO and -NO2 groups are not polar.
(B) they are aromatic compounds.
(C) intra molecular H-bond is present
(D) their molecular weights are high.
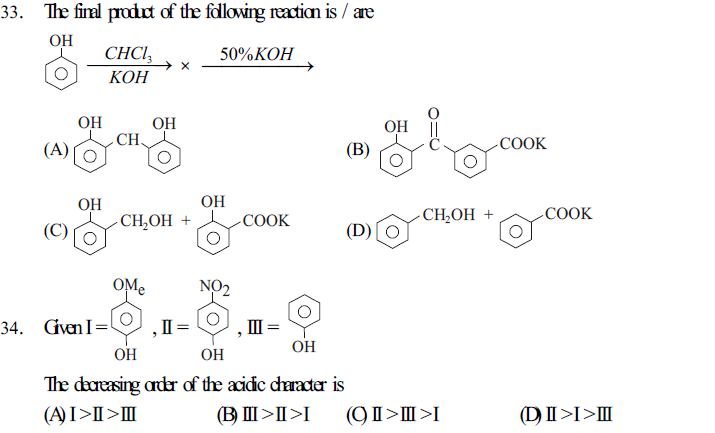
35. An organic compound `X' With molecular formula, C7 H8 O is insoluble in aqueous NaHCO3 but dissolves in NaOH . When treated with bromine water, `X' rapidly gives `Y' ( C7 H5 OBr3) The compounds `X' and `Y' respectively, are
(A) benzyl alcohol and 2,4,6 - tribromo-3-methoxy benzene
(B) benzyl alcohol and 2,4,6 - tribromo-3-methyl phenol
(C) o-cresol and 3,4,5 -tribromo-2- methyl phenol
(D) m-cresol and 2,4,6 -tribromo -3- methyl phenol
36. Willamson's Synthesis is used for the preparation of
(A) acid (B) ester (C) ether (D) alcohol
37. P - cresol reacts with chloroform in alkaline medium to give the compound A, which adds hydrogen cyanide to form the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is

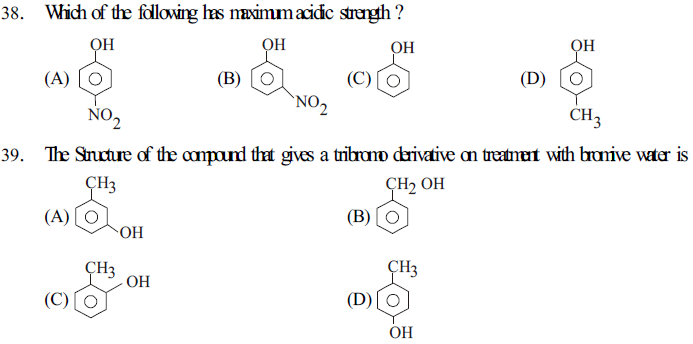
40. (I) Benzene 1,2 - diol (II) Benzene 1,3 -diol (III) Benzene 1,4 -diol (IV) Phenol
The incresing order of boiling points of above mentioned compounds is
(A) I < II < III < IV (B) I < II < IV < III
(C) IV < I < II < III (D) IV < II < I < III
41. Phenols are more acidic than alcohols because
(A) phenols are more soluble in polar solvents
(B) phenoxide ion is stabilised by resonance
(C) phonoxide ion do not exhibit resonace
(D) alcohols do not lose H atoms at all
42. The products obtained when benzyl phenyl ether is heated with HI in the mole ratio 1:1 are
1. phenol, 2 benzyl alcohol, 3. benzyl iodide, 4. iodobenzenc
(A) 1 and 3 only (B) 3 and 4 only (C) 1 and 4 only (D) 2 and 4 only
45. Which one of the following is reduced with zinc and hydrochloric to give the corresponding hydrocarbon?
(A) Ethyl acetate (B) Acetic acid (C) Acetamide (D) Butan -2-one
46. An organic compound x with molecular formula C5H10O yields phenyl hydrazone and gives a negative response to the iodoform test and Tollen's test. It produces n-pentane on reduction. The compound could be.
(A) petanal (B) pentan-2-one (C) pentan-3-one (D) amyl alcohol

54. Oxidation of is isopropyl benzene by oxygen in the presence of dilute acid gives___________.
(A) C6H5COOH (B) C6H5COCH3 (C) C6H5 CHO (D) C6H5 OH
55. Cross aldol condensation occurs between
(A) two same aldehydes (B) two same ketones
(C) two different aldehydes and ketones (D) two same acids
56. Pentan - 3 - one is not obtainel from
(A) 2,2- dichloro pentane (B) 3,3- dichloro pentane
(C) pentan -3- ol (D) pent -2- yne
57. C2H5CHO and ( CH3)2 CH CO be distiguished by testing with
(A) phenyl hydrazine (B) hydroxyl amine
(C) Fehilng Solution (D) sodium bisulphide
58. Which of the following has the most acidic hydrogen ?
(A) hexan-3-one (B) hexan 2,4- dione
(C) hexan 2,5-dione (D) hexan 2,3-dione

