DUAL NATURE OF MATTER AND RADIATIONS
ONE MARK QUESTIONS:
Question. An electron and alpha particle have the same de-Broglie wavelength associated with them. How are their kinetic energies related to each other?
Answer : 1. We have
λ= h/√2mk
or k=h2/2m λ 2 .
as λ e = λa
me< ma
so ke > ka
Question. The stopping potential in an experiment on photoelectric effect is 2 V. What is the maximum kinetic energy of the photoelectrons emitted?
Answer : 2. The maximum kinetic energy of the photoelectrons emitted = 1.6×10-19 ⨯ 2V
= 3.2× 10-19J
Question. Show graphically, the variation of the de-Broglie wavelength (λ) with the potential (V) through which an electron is accelerated from rest.
Answer : 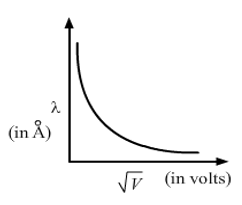
TWO MARKS QUESTIONS:
Question. In a photoelectric effect experiment, the following graphs were obtained between the photoelectric current and the applied voltage. Name the characteristics of the incident radiation (a) that was kept constant (b) that was variable in this experiment.
Answer : (a)Frequency of radiation
(b) Intensity of radiation
Question. A proton and an α-particle are accelerated through the same potential difference. Which one of the two has (i) greater de-Broglie wavelength, and (ii) less kinetic energy? Justify your answer.
Answer :

THREE MARKS QUESTIONS
Question. An electromagnetic wave of wavelength λ is incident on a photosensitive surface of negligible work function. If the photo-electrons emitted from this surface have the de-Broglie wavelength λ 1 , prove that λ =(2mc/h) λ21.
Answer : We have given work function W=0 ,so using Einestein photoelectric equation
1/2mv2=hν- W=hν
1/2mv2=hc/λ
Or, mv=√2mhc/ λ (1)
de -Broglie equation for emitted photoelectrons
λ 1=h/mv
using equation (1)
λ =(2mc/h) λ 21 (proved)
Question. Plot a graph showing the variation of stopping potential with the frequency of incident radiation for two different photosensitive materials having work functions w1 and w2 (w1 > w2). What is the significance of (i) slope and (ii) intercept of the lines depend?
Answer :
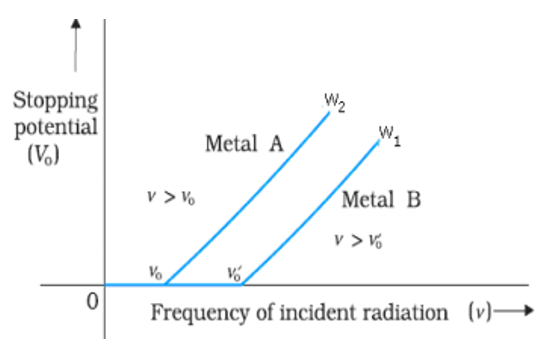
(a) From Einstein Photo electric equation the slope of this graph is h/e i.e. Planck constant divided by electronic charge.
(b) The intercept of this graph is W/e i.e. work function divided by electronic charge.
Question. (a) Why photoelectric effect cannot be explained on the basis of wave nature of light? Give reasons.
(b) Write the basic features of photon picture of electromagnetic radiation on which Einstein’s photoelectric equation is based.
Answer : Features of photoelectric effect cannot be explained on the basis of wave theory:
a) Maximum kinetic energy of emitted photoelectrons each independent of intensity of light.
b) There exists a minimum frequency i.e. threshold frequency for each photosensitive material below which no photo emission is possible.
c) Photoelectric effect is instantaneous in nature.
The properties of photon used to write Einstein’s photoelectric equation are
(i) The rest mass of the photon is zero.
(ii) The energy of the photon is E = hν.
Question. The given graph shows the variation of photo-electric current (I) versus applied voltage (V) for two different photosensitive materials and for two different intensities of the incident radiation. Identify the pairs of curves that correspond to different materials but same intensity of incident radiation.
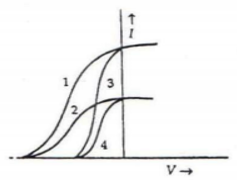
Answer : Curve 1 and 3 and curves 2 and 4 corresponds to different materials. For a given frequency of incident radiation the stopping potential is independent of intensity of light. As the pair of curves: 1&3 and 2 & 4 have different stopping potential so those materials are different.
1. If wavelength of electromagnetic waves are doubled what will happen to energy of photon?
2. Alkali metals are most suitable for photoelectric emission. Why?
3. Out of microwaves, UV, IR which radiation will be most effecting for emission of electrons from a metallic surface?
4. Can X-rays cause photoelectric effect?
5. If the intensity of incident radiation on a metal is doubled what happens to the K.E of electrons emitted?
6. What is the value of stopping potential between the cathode and anode of photocell? If the max K.E of electrons emitted is 5eV?
7. It is easier to remove an electron from sodium than from copper, which has a higher value of threshold wavelength?
8. What is the role of photocell in cinematography?
9. An electron and photon possessing same K.E. Which one will have greater wavelength?
10. In Davisson – Germer experiment if the angle of diffraction is 520 find Glancing angle?
11. What is the energy associated with a photon of wavelength 6000 A0 ?
12. What is the effect on the velocity photo electrons, if the wavelength of incident light is decreased?
13. Show graphically how the stopping potential for a given metal varies with a frequency of the incident radiation.
14. To work functions 2ev and 5ev for two metals x and y respectively. Which metal will emit electrons, when it is irradiated with light and wave length 400nm and why?
15. A photon and an electron have same de-broglie wavelength. Which has greater total energy.Explain?
16. The de-broglie wave length of a photon is same as the wave length of electron. Show that K.E. of a photon is 2mc ג/h times K.E. of electron. Where ‘m’ is mass of electron,c is velocity of light.
17. Derive the expression of de-broglie wave length in terms of energy of energy and temperature?
18. Light from bulb falls on a wodden table but no photon electrons are emitted why ?
If each metal is exposed to radiation of wavelength 300nm which of them will not emit photo electron.
20.An electron and alpha particle and proton have same kinetic energy , which have shortest De-broglie wavelength?
21. The De-broglie wave length associated with proton and neutron are equal.Which has greater kinetic energy?.
22.A stream of electron travelling with a speed at right angle to a uniform electric field E, is deflected in a circular path of radius “r” . Prove that e/m = v2/rE.
23.If the potential difference used to accelerate electron is doubled , by what factor the Debroglie wave length of the electron beam changed.
24. The De-broglie wave length associated with an electron accelerated through the potential difference “V” is λ. What will be its wave length , when accelerating potential is increased to 4v?
25.Visible light can not eject photo electrons from copper surface, whose work function is 4.4 ev , why? Prove mathematically.
EXTRA QUESTIONS
1. Neutrons, in thermal equilibrium with matter have an average KE = 3/2 kT .Compute de-Broglie’s wavelength associated
2. A nucleus of mass M initially at rest splits in two fragments of masses M/3 & 2M/3. Find the ratio of de-Broglie’s wavelength of two fragments.
3. X-rays of wavelength 0.82 A0 fall on metallic surface. Calculate de-Broglie’s wavelength of emitted photoelectrons. Ignore j of the metal.
4. Wavelength of photon and de-Broglie’s wavelength of electron has same value. Show that energy of photon is 2λmc/h times the KE of electron.
5. Compare energy of electron of de-Broglie’s wavelength 1 A0 with that of an X-ray photon of the same wavelength.
6. Calculate the ratio of de-Broglie wavelength associated with deutron moving with velocity ‘2v’ and a α-particle moving with velocity ‘v’.
7. An α-particle and proton are accelerated from rest through same PD ‘V’. Find the ratio of de-Broglie wavelength associated with them.
8. Calculate de-Broglie’s wavelength associated with an electron of energy 200eV. What will be the change in l if accelerating potential is increased to 4 times.
9. What is the (i) momentum (ii) speed and (iii) de-Broglie wavelength of electron of kinetic energy 120 eV.
10.Show that the wavelength of electromagnetic radiation is equal to the de-Broglie wavelength of its photon.
11. An electron, a-particle and a proton have same KE. Which of these particles have shortest wavelength?
12. The threshold frequency for a certain metal is 3.3*1014 Hz. If light of frequency 8.2*1014 Hz is incident on the metal. Predict the velocity of ejected electrons and cut-off voltage.
13.The work function of two metals A and B are respectively 1.2 eV and 2.4 eV. Light of wavelength 600nm falls on these metals.
(i) Which metal / metals will give photoelectric emission?
(ii) What is the maximum velocity and cut-off potential?
(iii) If the source is moved away, how does it affect the stopping potential?
14. The energy flux of sun reaching the earth is 1.3888*103 Wm-2. How many photons per square m is incident on the earth per sec. Assume average wavelength of sunlight = 550nm.
15. In an experiment on photoelectric effect, the slope of cut-off voltage versus frequency of light is found to be 4.12*10-15 Vs. Calculate the value of Plank’s constant.
16.The Photoelectric cut-off voltage for certain metal is 1.5V. What is the maximum KE of photoelectrons?
17. The threshold frequency of metal is ‘f’. When the light of frequency 2f is incident on it the maximum velocity of photoelectrons is ‘v1’. When the frequency is increased to 5f, the maximum velocity of photoelectrons is ‘v2’ Find v1 /v2.
18.The work function of cesium is 2.14 eV. Find (i) threshold frequency (ii) wavelength of light if the photoelectrons are stopped with stopping potential 0.6V.
19.In a plot of photoelectric current versus anode potential, how does a. the saturation current vary with anode potential for incident radiations of different frequencies but same intensity.
b. The stopping potential varies for incident radiations of different intensities but same frequency?
c. Photoelectric current vary for different intensities but same frequency of incident radiations? Justify your answer in each case.
20. The radiations of frequency 1015 Hz are incident on two photosensitive surfaces A and B. Following observations are recorded:
Surface A: no photo electric emission takes place.
Surface B: Photoemission takes place photo electrons have zero energy.
Explain the above observations on the basis of Einstein’s photoelectric
equations. How will the observations with surface B change when the wavelength of incident radiations is decreased?
21. An electron, α-particle and a proton have same de-Broglie wavelength.
Which of these particles has (i) minimum KE and (ii) maximum KE and why? In what way has the wave nature of electron beam exploited in electron microscope?
22.Calculate the (i) momentum and (ii) de- Broglie wavelength of electron accelerated through a potential difference of 56 V. On increasing the potential how this can improve the resolving power of a microscope.
23. Light of frequency 2.5 v0 is incident on surface of threshold frequency n0 and the photoelectric current is 1 mA. If frequency of light is halved and intensity is doubled, find new photoelectric current.
24. For what KE of neutron, will the associated de Broglie wavelength be 1.32*10-10 m?
25.The work function of three metals Na, K and Mo are respectively 1.92, 2.15 and 4.17 eV. If each of the metal is exposed to light of wavelength 300 nm, which of them will not emit photoelectrons and why?
26.By how much would the Stopping Potential a for given surface goes up if the frequency of the Incident radiation were to be increased from 4 ×1015Hz to 8 × 1015Hz?
27. Calculate de-Broglie wave length of
(i) an electron (mass 3 × 10 –2 kg moving with speed 100 m/s. Hence show that wave nature in hydrogen atom) moving with speed 1/100 of speed of light in vacuum and (ii) a ball of radius 5 mm and of matter is important at atomic level but is not really relevant at the macroscopic level.
28.The de-Brouglie wavelength associated with proton and a neutron is found to be equal. Which of the two has higher value of kinetic energy?
29. Graph showing variation of VS with frequency for two M1 M2 material is given.
(i) What are the values of work function for M1 and M2 VS
(ii)The values of VS for these for frequency v3 (v3 > v32)
are V1 and V2. Show that slope of lines equals
V1 - V2) / (V02 - V01)
30.Through what potential difference should an electron be accelerated so that its de-broglie wavelength becomes 0.4 Ao.
31.Monochromatic light of frequency 6x1014 Hz is produced by a LASER. The power emitted is 2x10-3 W. a) What is the energy of photon in the light beam. b) How many photons per sec on the average are emitted by the source. [Ans. (a) 2.49 eV, (b) 5x1015 sec-1]
32.If 5 % of the energy supplied to an incandescent light bulb is radiated as visible light, how many visible quantas are emitted by a 100W bulb? Assuming the wavelength of all the visible light to be 5000 Ao.[Ans. 1.41x1019 J]
33.If the wavelength of incident light changes from 4400 Ao to 4000 Ao, then find the change in stopping potential. [Ans. -0.26 eV]
UNIT 07
DUAL NATURE OF MATTER AND RADIATION
One Mark questions
1) According to the quantum theory, what happens when the intensity of light increases?
2) If a LASER of power 3.98MW produces a monochromatic light of energy 2.48eV, how many photons per second, on an average, are emitted by the source?
3) Can all photons from a monochromatic light source emit photoelectrons of same kinetic energy?
4) What is maximum frequency of X-rays produced by 30KV electrons?
5) A nucleus of mass M, initially at rest splits into two fragments of masses M’/3 and 2M’/3(M>M’). Find the ratio of de-Broglie wavelengths of two fragments.
6) What does the slope of the Graph between frequency v/s stopping potential represent?
7) What is the nature of graphical relation between frequency of incident radiation and the stopping potential?
8) On which factor the magnitude of saturation photoelectric current depends upon?
9) The work function of aluminium is4.2eV. If two photons each of energy 3.5eV strike an electron of aluminium sheet then what will be the speed of electrons?
10) Write down the rest mass of photon?
Two marks questions--
11. Calculate the number of photons in 6.62J of radiation energy of frequency 1012 Hz. Given h=6.62*10-34 Js.
12. When photons of energy hν falls on an aluminium plate (of work function Eo ),photoelectrons of maximum kinetic energy K are ejected.
If the frequency of radiation is doubled, find the maximum kinetic energy of the ejected photoelectrons.
13. If electron, proton and helium have same momentum, then write relation between de-Broglie’s wavelengths of the above particles.
14. The energy of a photon is equal to the Kinetic energy of proton. Let λ1 be the de-Broglie wavelength of the proton and λ2 be the wavelength of the photon. Find the ratio λ1/ λ2 in terms of energy ‘E’ of photon.
15. Draw the Graph which represents the variation of particle momentum and associated de-Broglie wave length?
Three marks questions--
16. Alkali metals are most suitable for photoelectric effect. Explain why?
17. Show that the product of the slope of the stopping potential versus frequency graph and the electronic charge gives the value of Planck’s constant.
18. When radiation of wavelength λ is incident on a metallic surface, the stopping potential is 4.8 volts. If the same surface is illuminated with a radiation of double the wavelength, then the stopping potential becomes 1.6 volts. What is the threshold wavelength for the surface?
19. A source of 25 watt emits monochromatic light of wavelength 6600Å. If efficiency for photoelectric emission is 3 %, then find the photoelectric current.
20. What is the De-broglie wavelength of a nitrogen molecule in air at 300K? Assume that the molecule is moving with the root mean square speed of molecules at this temperature. (Atomic mass of nitrogen is =14.0076u)
ANSWER KEY
1 MARKS ANSWERS
1. Number of the photons increases.
2. Number of the photons increases per second = total energy per second\ energy of the photons
= 3.98×10-3 j/sec
3.98×10-19 joules
=1016 photons/sec
3. No
4. E/h = v
5. 1:1 ; λ=h/p
6. h/e
7. A straight line.
8. intencity of the light.
9. Zero.
10. zero.
2 MARKS ANSWERS
11. n=e/r=1022
12. k=hi-E0
=>E0=hr-k
k! = h(2v)-E0=2hr-(hr-k)
K! = k+hr.
13. λp= λe= λhe
14. λ1=h/√2mE ; λ2=h/p=hc/E
λ1 /λ2=√E/c√2m
THREE MARKS
16. Work function is less for alkali metals.
17. proof evo=hv-hvo
differention
eΔvo=hΔvo
h=e(Δvo/Δv)=e×slope
18. Putting the data in photoelectric equation and taking ratios
λo=4λ
19. No of photons/sec = power/hv =pλ/hc
= 8.31 × 1019
Each photon ejects one electron.
No. of electrons ejected = η × 8.31 × 1019
N=24.93x1017
Photochemical current =Ne
=24.93x1017x1.6x10-19
=0.4Ampere
20. 1amu=1.66x10-27 Kg
mass of N2 molecule=2x14.0076x1.66x10-27 Kg
vrms=√3KT/m
=h/mvrms=h/√3KTm
=6.63x10-34/√3x4.649x10-26x1.38x10-23x300
=2.8x10-11m
=.028nm
VERY SHORT ANSWER TYPE QUESTIONS:-
DUAL NATURE OF MATTER AND RADIATIONS
1. An electron and photon have same wavelength. Which one of the two has more energy?
Relativistic energy of a particle, E= (m0c4+p2c2)½
Hence the electron has more energy than photon.
2. If wavelength of electromagnetic waves are doubled what will happen to energy of photon?
E = hνD
=hc/λ = E ∝ 1/λ energy of proton reduces to half.
3.Alkali metals are most suitable for photoelectric emission. Why? Alkali metals have too low work functions. Even visible light can eject electrons from them.
4. Out of microwaves, UV, IR which radiation will be most effecting for emission of electrons from a metallic surface?
UV are most effective since they have highest frequency hence more energetic.
5. If the intensity of incident radiation on a metal is doubled what happens to the K.E of electrons emitted? K.E of photons remains unaffected since they do not depend
6. What is the value of stopping potential between the cathode and anode of photocell? If the max K.E of electrons emitted is 5eV? Stopping potential V0 = Kmax/e = 5ev/e =5 V
7. It is easier to remove an electron from sodium than from copper, which has a higher value of threshold wavelength?
w0 = hν0 = hc/λ0
∴λ0 ∝ 1/ w0
Since sodium has lower work functions than copper it is easier for electron ejection. As it is lower work function, higher wavelength.
8. An electron and proton possessing same K.E. Which one will have greater wavelength?
1/2 mv2 = (m2 v2)/2m = p2/2m
λe > λp
electrons have greater De broglie wavelength than proton .
9. In Davisson – Germer experiment if the angle of diffraction is 520 find
Glancing angle?
θ = 90 - φ/2
= 90 – 52/2 =64°
10. What is the effect on the velocity photo electrons, if the wavelength of incident light is decreased?
KE of photoelectrons is given by Einstein’s photoelectric equation.
Ek =1/2 mv2
= hll- w0
V a 1/ √λ
As wavelength decreases velocity increases.
11. The stopping potential for some material is 1.2 V. What is the energy range of the emitted photoelectrons? The range of energies of the emitted photoelectrons will be from 0 to 1.2 eV.
12.The intensity of incident radiations in a photoelectric experiment is doubled. How the stopping is potential affected? The stopping potential will remain unaffected because it does not depend on the intensity of the incident light.
13. If the intensity of the incident radiation on a metal is doubled, what happens to the kinetic energy of the emitted photoelectrons? There is no change in the kinetic energy of the emitted electrons. This K.E. is independent of the intensity of the incident radiation as long as its frequency remains the same.
14. The frequency (υ) of incident radiation is greater than threshold frequency (υq) in a photocell. How will the stopping potential vary if frequency (υ) is increased, keeping other factors constant? When, (υ >υo) stopping potential will be increased.
15.What is the energy and wavelength of a thermal neutron ?
Kinetic energy of a thermal neutron = f/2 kBT. Since degree of freedom of a thermal neutron is three.
K.E = 3/2 kBT = 6.06 x 10-21 J
Wave length ,λe = h/(2mK.E)1/2 = , λe = h/(3m kBT)1/2=0.147nm.
16. An X –ray tube produces a continuous spectrum of radiation with its shots wavelength end 0.45 °A. What is the maximum energy of a photon in the radiation in electron volt?(ii) From your answer to (i) what order of accelerating voltage (fo0r electron) is required in such a tube?.
E = hu =hc/λ =27.6 x 103 eV
(ii)In this case energy of X-rays photon is 27.6 KeV, the striking electron must be of energy higher than 27.6 KeV. Therefore an accelerating voltage of the order of 30 KV is required.
17. It is difficult to remove free electrons from metal X as compare to metal Y . What you infer? Work function of metal X is higher than metal Y.
18. A particle behaves like a wave. What determine the wavelength of the wave? Momentum of the moving particle.
19. Draw a graph giving variation of maximum kinetic energy of photoelectrons with frequency of incident radiations. What is the slope of this graph?
The equation of this straight line is
K.E. = 1/2mv2max
= h (υ - υ0 )
20. The wavelength of radiations incident on a material is decreased. Does the maximum velocity of photoelectrons increase or decrease?
A decrease in wavelength implies an increase in frequency. Since an increase in frequency increases the maximum K.E. of the emitted photoelectrons, the maximum velocity would increase.
21. For photoelectric effect in sodium, the figure shows the plot of cut off voltage versus frequency of incident radiation. Calculate the threshold frequency (υ0) the work function for sodium
(ii)The work function (W) is related to the threshold frequency (υ0) by the relation.
Wo = hυo = 6.6 x 10-34 x 4.5 x 1014 J
= 29.7 xl0-20J = 2.97 xl0-i9J
= 2.97 x 10-19/1.6× 10-19 eV ≈1.856eV
22. Suppose the photoelectric effect occurs in a gaseous target rather than a solid. Will photoelectrons be produced at all frequencies of the incident photons?
No; we are likely to get photoemission only for those frequencies whose photons have an energy equal to or more than the (minimum) ionization energy for the gas concerned.
23. Yellow light does not eject photoelectrons from a given photosensitive surface, whereas green light does. What shall be situation in case of red and violet light?
We will not get any photoemission with red light since its frequency is less than that of yellow light. We will, however, get photoemission with violet light since its frequency is more than that of green light.
24. By what factor does the maximum velocity of the emitted photoelectrons change when the wavelength "the incident radiation is increased four times? (Given that the initial frequency used is five times the threshold frequency) When the wavelength is increased four times, the frequency goes down
1 mv12= h(ν1 −ν0) and 1/2 mv22= h(ν 2 −ν 0 )
Thus, the maximum velocity goes down by a factor of 4.
25. A cesium photocell, with a steady potential difference of 90 volt across it, is illuminated by a small bright light placed one metre away. The number of electrons that cross the photocell is n. What will be the number of electrons crossing the photocell when the same light is placed half metre away?
When the light is brought to a distance of 0.5 m, the intensity of the light falling on the photocell goes up four times. Since the number of photoelectrons emitted is directly proportional to the intensity of incident light, the new number of photoelectrons emitted would become 4n.
26. The sun rays are focused on a metal surface , and it produces a
current. The lens forming the image is then replaced by another lens of the same diameter but only half in focal length. What will be the effect on the photoelectric current?
Hence, the intensity of light falling on the metal surface gets reduced in case (b) and, we, therefore, get a reduced photoelectric current.
27. To work functions 2ev and 5ev for two metals x and y respectively. Which metal will emit electrons, when it is irradiated with light and wave length 400nm and why?
λ= 400 x 10-9m=4 x 10-7m
E= h c/λ = (6.6 x 10-34 x 3 x 108)/ (4 x 10-7) = 4.98 x 10-19 j
E= (4.98 x 10-19)/ (1.6 x 10-19) =3ev
Hence, metal x will emit electrons.
28. A photon and an electron have same de-broglie wavelength. Which has greater total energy. Explain ?
For a photon E1=hc/λ
For an electron λ=h/mv or m=h/λv
E2=mc2
= (h/λv) x c2
E2/E1=c/v>1
Therefore,E2>E1.thus, electron has total energy greater then that of photon.
29. The de-broglie wave length of a photon is same as the wave length of electron. Show that K.E. of a photon is 2mcλ/h times K.E. of electron. Where ‘m’ is mass of electron, c is velocity of light.
λph=λe=λ=h/mv
K.E. of photon Eph=hν=hc/λ
K.E. of electrons E=1/2mv2=1/2 m [h/mλ]2
=h2/2mλ2
Eph/Ee= (hc/λ) x 2mλ2/h2
=2mcλ/h
∴ Eph=Ee(2mcl/h)
30. How may photons are required for emission of one photo electron if frequency of incident radiation is less than threshold frequency. More than threshold frequency.
(i)No photo – electron will be emitted and photons are absorbed by electrons.
ii)One photon will emit one photo electron.
31. State the dependence of work function on the kinetic energy of electrons emitted in a photocell. If the intensity of incident radiation is doubled, what changes occur in the stopping potential and the photoelectric current?
According to Einstein theory of photoelectric effect, kinetic energy of emitted electron is
Ek = 1/2mV2 max
= hν −W0
Greater the work function of the metal, lesser the kinetic energy of the photoelectron. on doubling the intensity of the incident radiation stopping potential remains the same, whereas photoelectric current is doubled.
32. Using Davisson and Germer Experiment to establish the existence of de Broglie waves.
The experimental set up consists of an electron gun connected to a low tension battery. The electrons emitted by the gun are accelerated to a desired velocity by applying a suitable potential difference V using a high tension battery. Value of V can be varied. Using Bragg's law of diffraction of light through crystal lattices, the wavelength of the wave showing diffraction peak at 50° was calculated as 1.65 A.
The predicted de Broglie wavelength of electrons accelerated through 54 V is
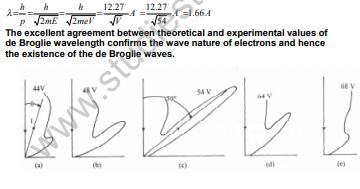
33. Through what potential difference an electron be accelerated so that it may have de – broglie wavelength 0.5A?
Let v = potential through which electron is accelerated.
Therefore, Energy = ½ mv2 = eV
=> λ =h/(2mE)1/2 = h/(2mev)1/2
=> v = h2/ 2meλ2
here, h = 6.625 × 10-34Js m = 9.1 × 10-31kg
e = 1.6 × 10-19 C
A =0.5 × 10-10 m hence v = 6.03 × 104 V
34. An e- and photon have the same energy of 100eV. Which has greater associated wavelength?
Here, Ae = h/(2mEe)1/2
Therefore the wavelength associated with a photon is greater than
an electron for the same energy.
35. the frequency of the light falling on the metal is doubled, what will be the effect on photocurrent & the maximum Kinetic Energy?
The photo current does not depend on the frequency of incident
radiation as:
36.e work function of ‘Li’ and ‘Cu’ are 2.3eV & 4eV. Which of these metals will be useful for the photoelectric cells working with the visible light?
The threshold wavelength of a metal is:
Thus, the incident trequency ,v is greater than v0 (Na) and v0 (K) but less than v0 (Mo) and v0 (Ni) , therefore Mo and Ni will not give photo electric emission.
If the laser is brought closer , intensity decreases. This does not effect the result regarding Mo and Ni but photoelectric current will increase for Na and K.
40. a) show that a free e- at rest can not absorb a photon and thereby acquire K.E. equal to the energy of the photon. Would the conclusion change if the e- has a constant velocity.
b) If the absorption of a photon by a free e- as proved in (a) , how does photo electric takes place?
Ans: a) the total energy of an e- , E = (m02c4 +p2c2 )1/2
= m0c2+h v
Squaring both the sides p2c2+2m0c2pc= p2c2
Or 2m0c2pc=0
This is impossible
b) we have shown in (a) that e-+y gives e-. however, for an e- a lattice the momentum of the incident photon can be shared by both the e- and the lattice while the lattice due to large mass does not share the energy of the photon. Thus, e- + y +lattice gives e- +lattice is not forbidden.
41. A monochromatic point source, S radiating wavelength 6000 Å with power 2W, an aperture A of diameter .1m are placed , area A = .5 cm2, efficiency = .9 , h = 6.63*10-34js, c= 3*108 , e= 1.6*10-9C .
calculate the photon flux at the center of the screen and photo current in the detector if a concave lens L of focal length .6m is inserted in the aperture find the new values of photon flux and photocurrent , uniform average transmission is 80% .If the work function of the photo emissive surface is 1eV , calculate the values of the stopping potential.
Sol : Energy of each photon , E = hc/ A =6.63*10-34*3*108/6*10-7 J
=3.315*10-19J
Therefore number of photon emitted= power / energy of each photon
= 6.03*1018s -1
Number of photons passing through the aperture of diameter .1m
NA = 6.03*1018s-1/4n (.6)2 *
n*(.05)2=1.047*1016s-1
Now the screen is at a distance of 6m from the source let the area of the screen iluminated by the light from be a, therefore, a/n* (0.5)2 or a=2.5πm2
The number ns = 1.33 *1016 photons Number of photoelectrons = .6*1012 electrons Photocurrent = .096 microA
b) 1/v = 1/f+ 1/u and thus, v= -.3m
photons transmitted through the lens n’ = .838* 1016 s-1
area uuum natea =a· = ..9025 πm2
photon flux = n’/a’= 2.95*1015 photons
therefore photocurrent = .012 microA
42: Standing distance of the waves = 2 Å , d increased to 2.5 Å . energy of the electrons = ? . least value of d for which wave is defined ?
Ans: nλ = a lTor stana ng wave)
Therefore n = 2d1/λ ana n+1 = 2d2/λ
Therefore n+1-n = 2d2/λ -2d1/λ
For least value of d, n is 1
Tnusλ= 2dmin
Energy of the electron E = p2/2me
Now p=n /λ
Therefore E= 150.95 eV

