Chapter-5
Surface Chemistry
Question. A colloidal solution can be purified by the following method :
(A) dialysis
(B) peptization
(C) filtration
(D) oxidation
Answer : A
Question. Which of the following is not a gel?
(A) Cheese
(B) Jellies
(C) Curd
(D) Milk
Answer : D
Question. An emulsion is a colloidal system of
(A) two solids
(B) two liquids
(C) one gas and one solid
(D) one gas and one liquid
Answer : B
Question. Following is the variation of physical adsorption with temperature:
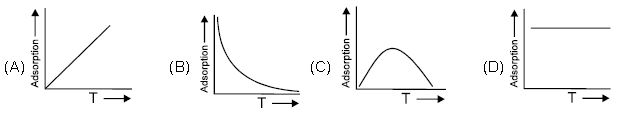
Answer : B
Question. Peptisation is :
(A) conversion of a colloidal into precipitate form
(B) conversion of precipitate into colloidal sol
(C) conversion of metal into colloidal sol by passage of electric current
(D) conversion of colloidal sol into macromolecules
Answer : B
Question. The nature of bonding forces in adsorption
(A) purely physical such as Van Der Waal's forces
(B) purely chemical
(C) both chemical and physical always
(D) none of these
Answer : D
Question. Reversible adsorption is
(A) chemical adsorption
(B) physical adsorption
(C) both
(D) none
Answer : B
Question. Which of the following ions is most effective in the coagulation of an arsenious sulphide solution ?
(A) K+
(B) Mg2+
(C) Al3+
(D) C
Answer : C
Question. The Tyndall effect associated with colloidal particles is due to
(A) presence of electrical charges
(B) scattering of light
(C) absorption of light
(D) reflection of light
Answer : B
Question. Which one of the following is not applicable to chemisorption?
(A) Its heat of adsorption is high
(B) It takes place at high temperature
(C) It is reversible
(D) It forms mono-molecular layers
Answer : C
Question. At CMC, the surfactant molecules :
(A) Decomposes
(B) Become completely soluble
(C) Associate
(D) Dissociate
Answer : C
Question. Which one is false in the following statement ?
(A) A catalyst is specific in its action
(B) A very small amount of the catalyst alters the rate of a reaction
(C) The number of free vacancies on the surface of the catalyst increases on sub-division
(D) Ni is used as a catalyst in the manufacture of ammonia
Answer : D
Question. Gelatine is generally added to ice creams. Why?
Answer: Ice cream is an emulsion of milk or cream in water (o/w type). Gelatine is generally added to act as emulsifiers to form the stable emulsion.
Question. Why is chemisorption called activated absorption?
Answer: For a chemisorption to take place ,reactants should acquire minimum energy of activation.
Question. Why is desorption necessary in catalytic reaction?
Answer: Desorption is necessary in catalytic reaction to maintain the flow of reaction.
Question. In chemical reaction can a product formed act as catalyst? If so give example.
Answer: Yes, Example: hydrolysis of ester. Ester hydrolysis is catalysed by acid. In the course of the reaction carboxylic acid is produced which catalyses the reaction.
Question. What is colloidion solution?
Answer: Colloidion solution is the solution which is used to impregnate the filter paper to make it an ultra filter.
Question. Give conditions for Tyndall effect to take place?
Answer: The refractive indices of dispersion medium & dispersed phase should not be same. The wavelength of the light should not be too low.
Question. Why is the ester hydrolysis slow in the beginning & becomes faster after sometime?
Answer: The ester hydrolysis slow in the beginning & becomes faster after sometime because the carboxylic acid produced acts as the catalyst.
Question. What are the reasons for charge on sol particles?
Answer: Frictional electrification Electron capture Preferential adsorption of ions from solutions Dissociation of surface molecules
Question. Why colloidal medicines are more effective?
Answer: Colloidal medicines have large surface area for the reaction. Hence colloidal medicines are more effective.
Question. Give the factors governing colour of colloidal solution?
Answer: Wavelength of light scattered, the manner in which light is received by observer, particles size.
Question. Why are Lyophylic colloids self stabilized?
Answer: Due to the force of attraction between dispersion medium & dispersed phase.
Question. State one application ZSM-5.
Answer: Used for the conversion of alcohol to gasoline.
Question. Explain the role of catalyst in a chemical reaction.
Answer: It lowers the activation energy & hence increases the rate of the reaction.
Question. Adsorption is always exothermic. Why?
Answer: During adsorption, the bonding takes place between adsorbate & adsorbent and there is a decrease in surface area & decrease in entropy.
Question. Define weeping of gels. Give the other name of weeping?
Answer: When gels are allowed to stand for a long time they give out small quantity of trapped liquid which accumulates on its surface. This action of gels is called as weeping, other name is syneresis.
Question. What is Thixotrophy?
Answer: Conversion of sol to gel is called thixotrophy.
Question. Why is FeCl3 preferred over KCl for a cut leading to bleeding?
Answer: Fecl3 has more positive charge and causes coagulation of blood quickly.
Question. Why is silica gel used as dehumidifier?
Answer: Silica gel adsorpes moisture on its surface quickly.
Question. Give the kinetics of adsorption of gases on metal surfaces.
Answer: Adsorption of gases on metal surfaces follows zero order kinetics.
Question. G is negative for adsorption process. Why?
Answer: Adsorption is exothermic & decrease in entropy occurs in adsorption.
Question. Give the significance of Brownian movement.
Answer: It gives stirring effect to the colloid. Hence colloidal particles do not aggregate. The colloid becomes stable.
Question. What happens when blood charcoal is shaken with conc. Kcl
Answer: in one case and its dilute KCl solution in other case? In conc. KCl positive adsorption occurs & in dilute Kcl negative adsorption occurs.
Question. Give the principle used in gas masks?
Answer: Competing adsorption
Question. Gold sol appears red , purple & also golden in colur Why?
Answer: Colour of the gold sol changes with its particle size.Its’ colour is red & becomes puple when size increases & finally becomes golden.
Question. Give the sign of ΔH & ΔS when Br2 gas gets adsorbed on charcoal.
Answer: ΔH = negative & ΔS = positive
Question. What is Argyrol?
Answer: Colloid of Ag with proteine(mild silver protein) Antiseptic & used for treating gonoria (a disease)
Question. What happens when NaCl is added to ferrichydroxide sol?
Answer: When NaCl is added to ferrichydroxide sol coagulation occurs.
Question. Differentiate dilute soap solution & concentrated soap solution.
Answer: Dilute soap solution behaves like a true solution & concentrated soap solution behaves like a colloid.
Question. Comment: Colloid is not a substance but it is a state of a substance.
Answer: The nature of the sub whether colloid or crystalloid depends upon the particle size.When the size of the solute particles lies between 1000 – 10000 pm , it behaves as a colloid & whenit is less than this value it behaves as a crystalloid. Thus Colloid is not a substance but it is a state of a substance which depeds upon the particle size.
Question. What happens when Fe(OH)3 is added to Gold sol?Why?
Answer: The Gold sol is negatively charged, Fe(OH)3 is positively charged & so coagulation occurs.Color of Gold sol changes from red to blue. Gold sol changes from red to blue due to the change in the particle size.
2 Marks Questions
1 Explain how activated charcoal adsorbs organic dye.
2. A graph between log(x/m) and log p is a straight line at angle of 450 with intercept on the y-axis( log k ) equal to 0.301. Calculate the amount of the gas adsorbed per gram of the adsorbent under a pressure of 0.4 Atmosphere
3 Adsorption is always exothermic in nature. Comment.
4 Critical temperatures of N2, CO, CH4 are 126, 134, and 110 K respectively. Arrange them in increasing order of adsorption on the surface of activated charcoal?
5.Consider the adsorption isotherms given below and interpret the variation in the extent of adsorption (x/m) when:
(i) Temperature increases at constant pressure.
(ii) Pressure increases at constant temperature
6.If the flocculation values of NaCl and AlCl3 are respectively 52 and 0.093, compare their coagulating powers.
7. Explain how soap solution stabilizes an emulsion of oil in water?
8.What happens when a freshly precipitated Fe(OH)3 is shaken with little dil. FeCl3 solution? Explain with possible reactions.
9.A methanol poisoned patient is treated by giving intravenous infusion of dil. ethanol. Explain. [Hint: Influence of inhibitors]
10.How does a ‘collector’ separate the ore from gangue in the froth floatation process?
3 Marks Questions
11 A colloidal solution of AgI is prepared by 2 different methods as shown:
(i) What is the charge of AgI colloidal particles in the two test tubes (A) and (B)?
(ii) Give reasons for the origin of charge.
12 SnO2 forms a positively charged colloidal sol in the acidic medium and negatively charged sol in basic medium. Explain.
13 1 g of charcoal adsorbs 100 ml of 0.5 M CH3COOH to form a monolayer and thereby the molarity of acetic acid is reduced to 0.49 M. Calculate the surface area of the charcoal adsorbed by each molecule of acetic acid. Surface area of charcoal = 3.01 × 102 m2/g.
14 To 100 ml of M/2 oxalic acid solution 2 g of active charcoal is added. After adsorption the strength of solution is reduced to M/4. Calculate the acid adsorbed by 1 g of charcoal.
15 Explain why:
(i) At sunset an orange colour develops in the sky.
(ii) Bleeding due to a small cut can be stopped by rubbing alum.
Activated charcoal is used in gas masks used by coal miners.
1 - MARK QUESTIONS
1. Why is it necessary to remove carbon monoxide when ammonia is obtained from Haber’s process? 1
2. How does BF3 act as a catalyst in industrial process? 1
3. The conductance of an emulsion increases on adding common salt. What type of emulsion is it? 1
4. Gelatin is generally added to ice-creams. Why? 1
5. What is syneresis or weeping of gels? 1
6. Most effective electrolyte causing the coagulation of negatively charged AS2S3 solution is a) MgCl2 b) KCl c) K3 [Fe(CN)4] d) Na2 SO4 1
7. Silicate garden is developed by setting of colored ions in the void of silicates. It is a case of -----------------. 1
8. What is purple of casius? 1
2 - MARKS QUESTIONS
9. Which of the following has minimum coagulating value? Why? 2
a) NaCl b) BaCl2 c) Al2 (SO4 )3 d) KCl
10. Critical temperatures of N2, CO and CH4 is 126, 134 and 190 K respectively. Arrange them in increasing order of adsorption on the surface of charcoal. Give reason. 2
11. The layer of fat in the pans used for manufacturing soaps can be removed by adding boiling washing soda solution. How will you account or it? 2
12. Why artificial rain can be caused by throwing common salt on the clouds? Explain. 2
13. Explain the curdling of milk when it is sour. 2
3 - MARKS QUESTIONS
14. In order to coagulate a fixed amount of As2S3 sol how will NaCl, MgCl2 and AlCl3 vary in their activity? Explain with the help of related ule.
15. A colloidal solution of AgI is prepared by two different methods. (A) AgNO3 solution is added to excess KI solution. (B) KI solution is added to excess AgNO3 solution. What is the charge on the Ag I colloidal particles in the two cases. Explain. 3
16. To the aqueous solution of a salt taken in a tube, a few drops of blue litmus solution were added. Ammonia solution was added dropwise to the red solution formed till it regained its blue color. The solution was then heated for sometime and the tube was left undisturbed. A blue mass was seen floating in colourless solution. How will you account for this? Identify the tentative salt. 3
17. The volume of nitrogen gas (measured at STP) required to cover a sample of silica gel with a mono-molecular layer is 129cm3/g of gel. Calculate the surface area per gram of the gel if each nitrogen molecule occupies 16.2 x 10-20 m2
1 - MARK QUESTIONS
1. Why is it necessary to remove carbon monoxide when ammonia is obtained from Haber’s process? 1
2. How does BF3 act as a catalyst in industrial process? 1
3. The conductance of an emulsion increases on adding common salt. What type of emulsion is it? 1
4. Gelatin is generally added to ice-creams. Why? 1
5. What is syneresis or weeping of gels? 1
6. Most effective electrolyte causing the coagulation of negatively charged AS2S3 solution is a) MgCl2 b) KCl c) K3 [Fe(CN)4] d) Na2 SO4 1
7. Silicate garden is developed by setting of colored ions in the void of silicates. It is a case of -----------------. 1
8. What is purple of casius? 1
2 - MARKS QUESTIONS
9. Which of the following has minimum coagulating value? Why? 2
a) NaCl b) BaCl2 c) Al2 (SO4 )3 d) KCl
10. Critical temperatures of N2, CO and CH4 is 126, 134 and 190 K respectively. Arrange them in increasing order of adsorption on the surface of charcoal. Give reason. 2
11. The layer of fat in the pans used for manufacturing soaps can be removed by adding boiling washing soda solution. How will you account for it? 2
12. Why artificial rain can be caused by throwing common salt on the clouds? Explain. 2
13. Explain the curdling of milk when it is sour. 2
3 - MARKS QUESTIONS
14. In order to coagulate a fixed amount of As2S3 sol how will NaCl, MgCl2 and AlCl3 vary in their activity? Explain with the help of related rule.

